Dr. Trigo, José Manuel
Direktor für Onkologie, Forschung und Innovation
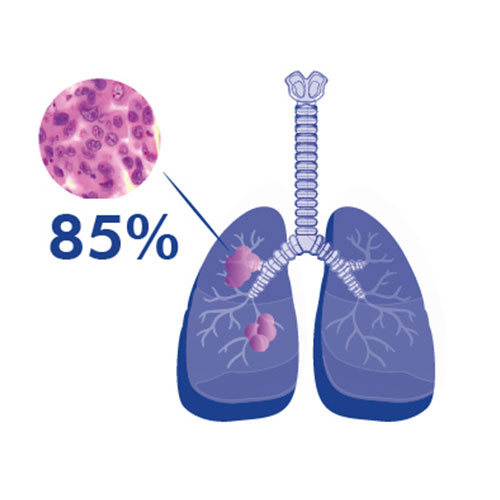
Non-small cell lung cancer is the most common type of lung cancer, accounting for 85% of cases.
Within this there are several subtypes, the most common are:
Depending on the type of tumour and each patient’s individual condition, your oncology team will propose different treatment options.
If there is no contraindication due to concurrent disease, and the patient has good lung capacity, surgery is the treatment of choice at these stages.
The goal of surgery is the complete removal of the lung tumour and lymph nodes near the tumour, as well as those in the mediastinum. The tumour should be removed with a surrounding healthy lung border (margin).
A „negative margin“ means that when the pathologist examines the lung or the part of the lung removed by the surgeon, they do not find traces of cancer in the healthy tissue surrounding the tumour. The affected lobes may be removed (lobectomy), but the entire lung may sometimes need to be removed (pneumonectomy).
If, for any reason, the surgeon cannot remove the entire lobe of the lung, they may perform a wedge resection of the tumour, with a surrounding normal lung margin.
After surgery, a decision is made on the indication for adjuvant (following surgery) chemotherapy. Your oncologist will make this decision after assessing all the information available.
Lymph node involvement and tumours of at least 4 centimetres in size are usually susceptible to chemotherapy, consisting of a combination of a drug called cisplatin with another chemotherapy drug.
Chemotherapy drugs are given through a vein every 21 days (known as 1 cycle) to try to destroy cancer cells that may remain in the body after surgery. If there is no toxicity, 4 cycles of adjuvant chemotherapy are usually given.
Sometimes chemotherapy is given prior to surgery (neoadjuvant chemotherapy) with the intention of reducing the size of the tumour. This is especially indicated in patients with stage III cancer where resection is questionable or where the mediastinum is involved, and chemotherapy is required prior to assessing whether or not further surgical treatment can be performed.
Patients with stage I to III disease who are not candidates for surgery due to other existing conditions, but who have good lung capacity, may benefit from radical radiotherapy treatment.
Radiotherapy treatment offered early is known as SBRT (Stereotactic Body Radiotherapy). This type of treatment requires very advanced, precise technology, as high doses of radiation must be delivered to the treatment zone to be effective. Due to its proximity to other organs it is a delicate area, prevention of damage to surrounding healthy tissue is therefore essential.
At HC Cancer Center we have the latest generation Tomotherapy using Synchrony. This adapts to respiratory movement during treatment delivery, increasing accuracy and preventing side effects as much as possible whilst helping to maintain respiratory capacity after treatment.
SBRT radiotherapy is a painless, minimally invasive treatment which is administered on an outpatient basis and usually requires between 1 and 5 sessions depending on the case.

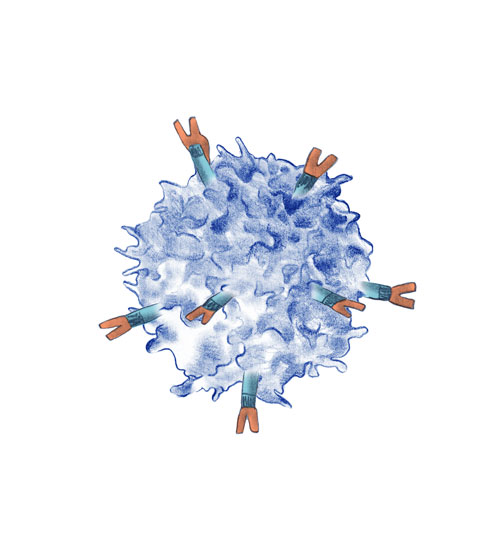
One of the reasons malignant cells succeed in growing is because they are able to bypass the immune system. Immunotherapy is a type of treatment which helps the immune system to fight cancer.
Clinical trials are investigating the role of immunotherapy in the initial stages. Drugs are administered intravenously which attempt to awaken the immune system so that it can act against the cancer.
There are different clinical studies into immunotherapy after surgery (adjuvant) or before surgery (neoadjuvant) in combination with chemotherapy. If your doctor proposes one of these studies, it may be an opportunity to test very promising treatments.
The main contraindication to receiving these treatments is in patients with autoimmune disease (e.g. Crohn’s disease, ulcerative colitis, rheumatoid arthritis).
The side effects are fewer than those from chemotherapy (tiredness, inflammation of the liver, effect on thyroid function, itchy skin, among others), they are usually mild and controlled by discontinuation of treatment and administration of cortisone.
For patients with locally advanced lung cancer who are not suitable for radical surgical treatment, the treatment of choice is chemotherapy with radiotherapy (if functional tests and patients’ other existing conditions allow).
This combination of treatment can be administered concurrently (at the same time) or sequentially (chemotherapy first followed by radiotherapy). If there are no contraindications, concurrent administration is preferable. Combined chemo-radiotherapy treatment lasts about 7 weeks, sequential treatment lasts around 12 weeks
Once chemo-radiotherapy is completed, if there is no progression (tumour growth), consolidation treatment with immunotherapy will be administered for one year.
In order to receive this treatment, the tumour must express a biomarker (test requested at biopsy). There must be PD-L1 expression of ≥ 1%, as this indicates that the tumour has some susceptibility for immunotherapy to be effective.

In patients with stage IV or advanced disease (patients with metastases), it is especially important for the oncologist to have biomarkers available before treatment is commenced as these are required to decide on the treatment approach.
To obtain these, your oncologist will usually ask for:
In some cases, your oncologist may request other biomarkers.
Depending on biomarker expression, your oncologist may prescribe immunotherapy treatment.
Immunotherapy may be given as a single or combined treatment with chemotherapy. It is given once every 3 weeks for up to a maximum of 2 years if there is no disease growth.
Chemotherapy with immunotherapy is given once every 3 weeks (1 cycle) with up to a maximum of 4 cycles. If after 4 cycles of combined treatment there is no tumour growth, you will receive maintenance treatment of immunotherapy and chemotherapy for up to 2 years if you have adenocarcinoma.
If patients have any contraindication to receiving immunotherapy, chemotherapy treatment is given exclusively for 4 to 6 cycles. If there is no disease progression it may or may not be followed by maintenance treatment, depending on the cancer subtype.
These drugs are taken orally every day until the tumour grows.
The main side effects are tiredness, diarrhoea, and skin spots, but for most patients this treatment is more comfortable and has fewer side effects than chemotherapy.
Genetic abnormalities are being increasingly identified in non-small cell lung cancer, these may be determinants for targeted treatment (RET, MET exon 14, NTRK…). A massive sequencing genomic test is now therefore recommended (Next Generation Sequencing or NGS).
Dr. Trigo, José Manuel
Direktor für Onkologie, Forschung und Innovation
Dr. Aguilar PerezGrovas, Ricardo
Neumology Specialist
Dr. Sedano Ferreras, Paula
Spezialist für Radioonkologie
Dr. García Baltar, José Antonio
Especialista en Radiofísica Hospitalaria
Dr. De Castro, Francisco Javier
Facharzt für Radiologie und Innere Medizin
Tel.: +34 952 908 628
+34 609 148 799
952908898 Onkologie
951829978 Bildgebende Diagnostik
951829947 Gynäkologie
952908897 Fertilitäts-Zentrum
951829947 Physiotherapie